Reducing Uncertainty in Behavioral Health Study Participation
Behavioral health studies are a critical part of clinical research. Unlike drug trials, these studies often focus on understanding and changing health behaviors, such as physical activity, sleep, substance use, or diet.
For children and adolescents, these studies can feel unfamiliar and difficult to understand. When youth are unsure what participation involves, they may feel hesitant about deciding to take part. For research teams, this uncertainty can impact recruitment and retention rates.
Demystifying Behavioral Studies for Youth
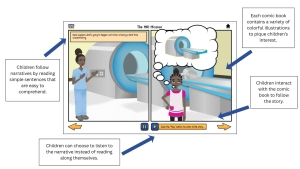
Behavioral health studies often involve concepts that are abstract for young people. Youth may not understand what health behaviors are, why they are being studied, or what participation will look like. Without clear explanation, these studies can feel vague or confusing.
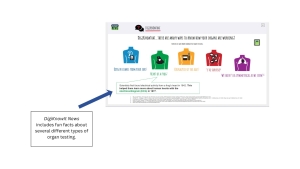
DigiKnowIt News introduces behavioral health concepts in ways that are clear and relatable for youth.
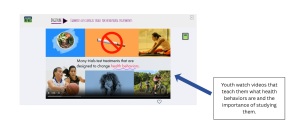
Helping Potential Participants Understand What Participation Looks Like
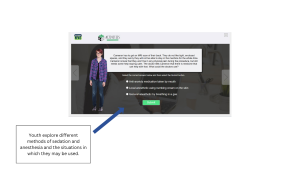
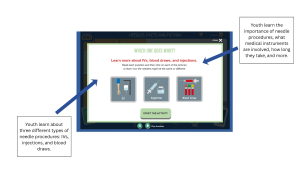
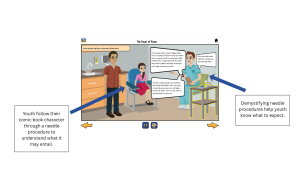
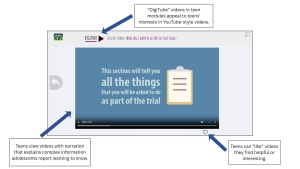
One of the sources of hesitation for assenting is not knowing exactly what participation involves. Behavioral studies may include surveys or questionnaires, tracking behaviors over time, and participating in interventions like counseling or programs. When these elements of a study are not clearly explained, youth may feel uncertain about what they are agreeing to, which could cause them to decline participation or withdrawal after assent.
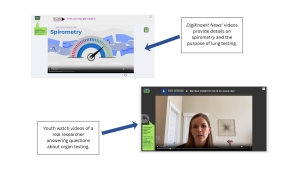
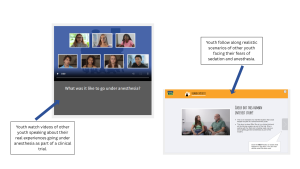
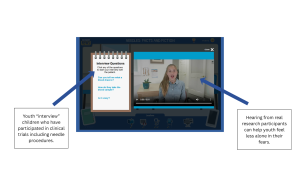
Hearing directly from other youth can help make participation feel more concrete. DigiKnowIt News incorporates video interviews with real youth to help children and teens visualize participation and feel seen in their fears or confusion.

Providing Opportunities to Explore How Behavioral Studies Work
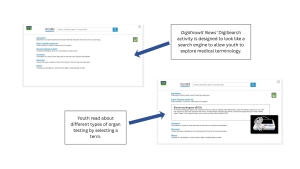
Beyond understanding participation, youth may also benefit from understanding how these studies are designed. DigiKnowIt News’ interactive activities allow youth to explore how behavioral studies are structured.
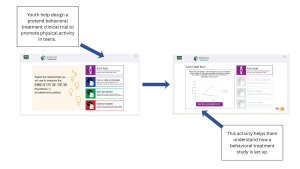
By helping “design” a behavioral clinical trial, youth learn about hypotheses, control groups, and how data is collected. Through this activity, youth learn how researchers test interventions, how data is collected and compared, and how different measures (like trackers or questionnaires) are used. This helps make the research process more transparent and less intimidating.
Building Trust in Youth for Research
Two important factors in reducing uncertainty to participate is providing youth with access to credible information and reducing their mistrust in research teams. DigiKnowIt News connects youth with real researchers to provide reliable and trustworthy information and increase their comfortability with receiving information from research teams.
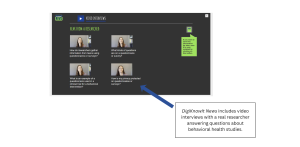
Support Behavioral Study Readiness with DigiKnowIt News
When youth better understand behavioral health studies, they are more prepared to make informed decisions about participation. Preparation can help reduce uncertainty, increase comfort with participation, and improve engagement throughout the study.
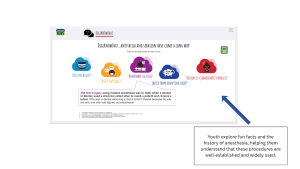
DigiKnowIt News prepares youth for behavioral health research through clear explanations of health behaviors and study goals, real-world examples of how studies are conducted, interactive learning experiences, and peer and researcher perspectives.
Learning activities about behavioral health studies are optional and included in the teen version of DigiKnowIt News, allowing research teams to tailor DigiKnowIt News content to their study.
Start providing clear, developmentally appropriate preparation to help reduce uncertainty and support more informed participation decisions in your study. Explore DigiKnowIt News at digiknowit.com.